CME
Article
Psychiatric Times
Rehabilitation Through Stimulation: Exploring Noninvasive Brain Stimulation for Substance Use Disorders
Author(s):
In this CME article, review the current treatment modalities for noninvasive brain stimulation in the treatment of substance use disorders, and to explore the potential for further expansion of indications.
PhotoGranary/AdobeStock
CATEGORY 1 CME
Premiere Date: January 20, 2024
Expiration Date: July 20, 2025
This activity offers CE credits for:
1. Physicians (CME)
2. Other
All other clinicians either will receive a CME Attendance Certificate or may choose any of the types of CE credit being offered.
ACTIVITY GOAL
To review the current treatment modalities for noninvasive brain stimulation in the treatment of substance use disorders, and to explore the potential for further expansion of indications.
LEARNING OBJECTIVES
1. Understand the role of noninvasive brain stimulation in the treatment of substance use disorders.
2. Become more familiar with the current treatment modalities and potential for further expansion of noninvasive brain stimulation indications for substance use disorders.
TARGET AUDIENCE
This accredited continuing education (CE) activity is intended for psychiatrists, psychologists, primary care physicians, physician assistants, nurse practitioners, and other health care professionals who seek to improve their care for patients with mental health disorders.
ACCREDITATION/CREDIT DESIGNATION/FINANCIAL SUPPORT
This activity has been planned and implemented in accordance with the accreditation requirements and policies of the Accreditation Council for Continuing Medical Education (ACCME) through the joint providership of Physicians’ Education Resource®, LLC, and Psychiatric Times®. Physicians’ Education Resource, LLC, is accredited by the ACCME to provide continuing medical education for physicians.
Physicians’ Education Resource, LLC, designates this enduring material for a maximum of 1.5 AMA PRA Category 1 Credits™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
This activity is funded entirely by Physicians’ Education Resource, LLC. No commercial support was received.
OFF-LABEL DISCLOSURE/DISCLAIMER
This accredited CE activity may or may not discuss investigational, unapproved, or off-label use of drugs. Participants are advised to consult prescribing information for any products discussed. The information provided in this accredited CE activity is for continuing medical education purposes only and is not meant to substitute for the independent clinical judgment of a physician relative to diagnostic or treatment options for a specific patient’s medical condition. The opinions expressed in the content are solely those of the individual faculty members and do not reflect those of Physicians’ Education Resource, LLC.
FACULTY, STAFF, AND PLANNERS’ DISCLOSURES AND CONFLICT OF INTEREST (COI) MITIGATION
None of the staff of Physicians’ Education Resource, LLC, or Psychiatric Times or the planners or the authors of this educational activity have relevant financial relationship(s) to disclose with ineligible companies whose primary business is producing, marketing, selling, reselling, or distributing health care products used by or on patients.
For content-related questions, email us at PTEditor@mmhgroup.com; for questions concerning the accreditation of this CME activity or how to claim credit, please contact info@gotoper.com and include “Rehabilitation Through Stimulation: Exploring Noninvasive Brain Stimulation for Substance Use Disorders” in the subject line.
HOW TO CLAIM CREDIT
Once you have read the article, please use the following URL to evaluate and request credit: https://education.gotoper.com/activity/ptcme24jan. If you do not already have an account with PER®, you will be prompted to create one. You must have an account to evaluate and request credit for this activity.
The emergence of neurostimulation undoubtedly has shaped clinical practice. Through the localized application of electrical or magnetic fields, clinicians can now directly modulate target neurocircuits to facilitate or inhibit activity. Noninvasive brain stimulation (NIBS) techniques, such as transcranial magnetic stimulation (TMS) and transcranial direct current stimulation (tDCS), are considered cutting-edge innovations within interventional psychiatry. A growing body of evidence shows efficacy in the treatment of a range of mental illnesses1 and promise in some unrelenting disorders. Repetitive TMS (rTMS) received US Food & Drug Administration (FDA) approval in 2008 as a clinical intervention for treatment-resistant depression (TRD).2
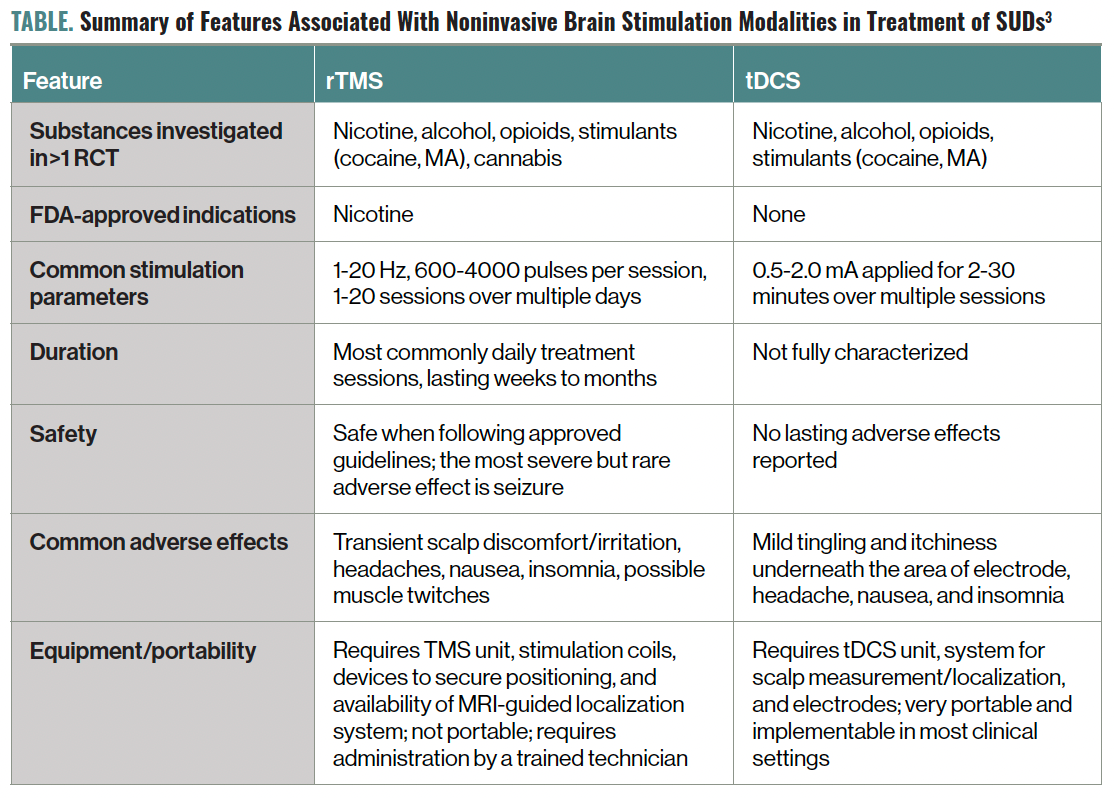
More recently, there exists burgeoning interest in the clinical use of NIBS in the treatment of substance use disorders (SUDs; Table).3 The FDA has cleared a novel TMS system to assist in smoking cessation4—a landmark event bringing recognition to the utility of NIBS for SUDs. Although complex psychosocial factors are involved in the development of SUDs, advancements in neuroscience have asserted a brain-based model of addiction. Chronic, recurrent drug use causes maladaptation to neural circuitry involved in cognition and emotional regulation.5 This can be experienced as increased impulsivity and intensifying motivation to seek and use the substance, further reinforcing habitual abuse and craving.5 By inducing changes in the dysregulated circuitry underlying addictive behavior, NIBS is theorized to promote recovery.3
Table. Summary of Features Associated With Noninvasive Brain Stimulation Modalities in Treatment of SUDs3
Types and Utilities of NIBS
Although the exact mechanisms of action are different, both induce neuronal activity in the brain region below the placement of the device on the scalp. Local cortical stimulation is thought to transmit to connected secondary brain networks, allowing indirect manipulation of network activity and enhancement of neuroplasticity.3
The standard unihemispheric format for tDCS involves the placement of 2 electrodes: a target placed on the scalp (the region corresponding to the cortical “target” below) and a reference electrode. A constant, low-intensity current flows to form a circuit through the cortical area between each.6 Positive anodal current flow is theorized to increase cortical activity, whereas negative cathodal current decreases6 cortical activity, which translates to enhancement or inhibition of the associated functions of the affected region.7
Newer formats include options for bihemispheric stimulation, the use of multiple smaller electrodes,7 and transcranial alternating current stimulation (tACS), which can modulate rhythmic waves of brain activity.6 Stimulation parameters are heterogeneous across settings; however, they commonly employ current intensities of 0.5 to 2.0 mA for 10 to 20 minutes per session for addictive disorders.6 Early evidence suggests stimulation should be at a minimum of 0.017 mA/cm2 to facilitate neuromodulatory effects,8 and effects increase along with the current density. However, other studies contradict a linear relationship,9 and one meta-analysis found no difference between 1 versus 2 mA current intensities when applied to SUDs.10
In contrast, TMS uses rapid, alternating magnetic pulses delivered through an electromagnetic coil to stimulate cortical activity at a selected brain site. Repetitive stimulation (ie, rTMS) is believed to enhance neuroplasticity locally and to affect connected dysregulated circuitry that may facilitate the maladaptive behaviors that underly disorders.11
Coils typically deliver between 600 and 4000 magnetic pulses per session in fixed or bursting patterns. Low frequencies (<1 Hz) will inhibit whereas high frequencies will excite underlying cortical excitability and neural activity.12 The appropriate dose of rTMS to administer is determined as a percentage value of the patient’s resting motor threshold. (For example, the smallest intensity produces a motor-evoked potential.)
The coil type determines the area and depth of stimulation. The common figure-of-8, circular, and H-coils are predicted to cover a surface area of approximately 10 cm2, 50 cm2, and 100 cm2, respectively.13 Although figure-of-8 and circular coils are estimated to penetrate 1 to 2 cm2, the newer H-coil extends 2 to 3 cm2, allowing localization of deeper subcortical structures, known as deep TMS (dTMS).14
Aberrant functioning in prefrontal brain networks holds clinical importance in the maintenance of SUDs.15 Particularly, the dorsolateral prefrontal cortex (DLPFC) is a focus of the larger-scale executive network implicated in drug-seeking behaviors and cravings.6,16 SUD severity has shown a correlation with the hypoactivation of this network across drug types.16 Currently, the DLPFC is the most common site of stimulation with the greatest clinical evidence.3,17
In clinical settings, treatment efficacy is typically measured by changes in the level of craving (often self-reported and/or following exposure to a drug-related cue) or consumption (or abstinence) from baseline for the drug of choice.3 Altogether, both tDCS and TMS show promising therapeutic potential in various drug-dependent populations. In one meta-analysis analyzing both tDCS and TMS studies targeting the DLPFC, a medium effect size was found in the reduction of craving levels across substance types.17
In clinical studies of tDCS, DLPFC stimulation has shown significance in reducing craving in populations with alcohol,18-22 nicotine,23-27 and cocaine use disorders.28 Outcomes in substance consumption are less consistent.29 Abstinence may improve concomitantly with perceived craving24,25,30; however, other studies report no change26,27 or reduced consumption without change in craving.30
Clinical trials of DLPFC stimulation by TMS have provided evidence in decreasing use of tobacco,4,31 alcohol,32 and stimulants including cocaine33 and methamphetamine.17,34-36 These substances are the most well-studied types of SUDs in rTMS trials.3 Notably, treatment of tobacco use disorder with the dTMS coil has been demonstrated in a pivotal large multisite, sham-controlled, randomized controlled trial, which led to FDA clearance of the device for this indication.4 Therefore, rTMS treatment of tobacco use disorder is likely the most readily translatable to routine clinical practice at this point, closely followed by alcohol and cocaine use disorders as possible emerging indications.
More studies are needed in the treatment of opioid use disorder and cannabis use disorder (CUD). Reductions in craving following DLPFC stimulation have been reported in patients with heroin34,37-40 and morphine41 dependence, as well as in patients with chronic pain who are taking prescription opioids.42 In the few trials on CUD, DLPFC stimulation decreased cannabis consumption and craving after multiple sessions,43,44 but not with single sessions44 or with stimulation directed to the parietal cortex.45
Recovery from addiction extends beyond eliminating drug use; assessing off-target effects in mood/anxiety, cognitive performance, sleep, and other behavioral changes can also help inform therapeutic outcomes.46 Some neurostimulation studies have investigated substance-dependent patients with comorbid psychiatric illnesses,29 which may share similarities in neurocircuitry dysregulation. Stimulation protocols in the treatment of SUDs are similar in design to those for TRD.47 In one example, rTMS treatment improved both depressive symptoms and perceived alcohol craving in a sample population with both major depressive disorder and alcohol use disorder.48
Clinical studies on tDCS and rTMS typically have small sample sizes and lack homogeneity across patients, which may make it difficult to interpret results as being reliable.49 Methodological inconsistencies and limitations further make firm conclusions of evidence difficult.49 The stimulation and treatment parameters that provide optimal outcomes have yet to be established.10
However, there is some emerging evidence on factors that heighten positive outcomes. Meta-analyses exploring the effects of NIBS treatment on self-reported craving and consumption across multiple substance abuse types demonstrate that having multiple sessions over different days improves efficacy, suggesting a cumulative effect10,17,36 and similarly longer durations of stimuli or several pulses in total.10,36 Additionally, no differences were found between right and left anodal placement,10,26 targeting the left versus right DLPFC, or in study design.10,17,36 Importantly, high-frequency (5-20 Hz) rTMS, as opposed to low frequency (approximately 1 Hz), is most frequently used and demonstrates the greatest effectiveness.36,50
Effects of stimulation can be variable to patients and setting factors. The baseline state of the individual is emerging as an important moderator of treatment efficacy,51 such that behavioral priming immediately before or during stimulation enhances effects.52 The FDA approval for rTMS use in OCD requires exposure to a stimulus related to the individual’s obsessions. In one large clinical trial applying rTMS to patients who smoke cigarettes, individuals exposed to a visual smoking cue before treatment displayed the greatest reduction in consumption and craving.31 Although this is promising, only approximately 40% of tDCS/rTMS studies incorporated cue exposure.3 Physiological states (eg, levels of alertness or caffeine intake53) may also influence outcomes, as well as individual differences.50 Some evidence suggests that women may show greater excitability to anodal tDCS,54 and in some studies assessing cognitive enhancement, effects differ by age.50
Each modality has existing guidelines that help guide patient selection. Both tDCS and TMS share similarities in exclusion criteria, which largely revolve around minimizing the risk of seizure.55 A thorough evaluation of histories of epilepsy, seizure, stroke, brain injury, and the presence of metal implants is pertinent. Many clinical studies exclude patients with neurological or other psychiatric diagnoses, and some evidence suggests those who have received these diagnoses are at higher risk for adverse effects.56 Although one study on tDCS demonstrated no harm to the fetus,57 NIBS generally should be avoided in patients who are pregnant, as safety has yet to be established. It is important to note that clinical considerations may further change as this field evolves.55
NIBS is generally considered safe and well tolerated, making this technology attractive for clinical implementation. One study of tDCS found that transient, mild adverse effects occur in an estimated 17% of healthy individuals.56 The most commonly reported adverse effect is a cutaneous sensation at the site of stimulation upon initiation,56 but other adverse effects, such as headache, fatigue, nausea, inattentiveness, and mood changes, may be possible.55,56,58 However, many studies may not report adverse effects, and there is a need for more standardized reporting and assessment.58
There has not been any indication that patients with addiction are at increased risk of adverse events or need to adjust parameters. However, any condition that may lower the threshold for seizures should warrant caution, and stimulation should be avoided in this state (eg, patients withdrawing from alcohol or benzodiazepines and those who are under the influence of cocaine or other stimulants). Ideally, patients should refrain from substance consumption for a reasonable period before stimulation sessions.55 In this sense, the timing of stimulation based on the patient’s position in substance recovery is important to consider.
Patients should be assessed for concurrent risky behaviors and motivation.3,59 The detoxification period involves a higher risk of relapse with heightened craving and withdrawal. NIBS can help alleviate these feelings during this vulnerable period.17 In contrast, the role of NIBS in sustaining long-term abstinence is limited.3 NIBS combined with behavioral and/or pharmaceutical interventions may further help reduce the risk of relapse later on and should be explored in further research.17,60 For example, one study combining rTMS with nicotine replacement therapy surprisingly had a nearly 50% abstinence rate.61
Concluding Thoughts
Before health care systems can adopt neurostimulation as a treatment option, clinical researchers must have a consensus on optimal stimulation parameters, treatment protocols, and patient selection, which evidence suggests also varies by substance type. Treatment of tobacco use disorders with rTMS has recently been cleared by the FDA and may represent the most clinically relevant indication for rTMS and SUDs at this point. Importantly, as this field grows in recognition, there is a need to increase training (or otherwise awareness) among technicians, prescribers, and clinicians; promotion within hospital infrastructure; and expansion in insurance coverage.
Although neurostimulation may initially appear to be an obscure therapeutic approach to psychiatrists and other clinicians who treat addictions, the scientific evidence is escalating, and clinicians may soon see these treatment modalities ready for translation to clinical practice.
Ms Goud is a medical student at the Temerty Faculty of Medicine at the University of Toronto in Ontario, Canada. She works as a national sustainability manager/project coordinator with the Mood Disorders Society of Canada, and her research interests focus on addictions and digital applications in mental health.
Dr Tang is an addiction psychiatrist and clinician scientist at the Centre for Addiction and Mental Health and an assistant professor in the Department of Psychiatry at the University of Toronto in Ontario, Canada. Dr Tang’s current focus is on the development of noninvasive brain stimulation treatments in addiction psychiatry, innovative approaches to evidence-based treatment delivery, and conducting clinical trials to investigate and target putative neurobiological mechanisms underlying these disorders. Dr Tang receives research support through the Brain & Behavior Research Foundation, the Physicians’ Services Incorporated Foundation, the Research in Addiction Medicine Scholars Program from the National Institute on Drug Abuse, and the Labatt Family Network for Research on the Biology of Depression.
References
1. Hyde J, Carr H, Kelley N, et al. Efficacy of neurostimulation across mental disorders: systematic review and meta-analysis of 208 randomized controlled trials. Mol Psychiatry. 2022;27(6):2709-2719.
2. Janicak PG, Nahas Z, Lisanby SH, et al. Durability of clinical benefit with transcranial magnetic stimulation (TMS) in the treatment of pharmacoresistant major depression: assessment of relapse during a 6-month, multisite, open-label study. Brain Stimul. 2010;3(4):187-199.
3. Ekhtiari H, Tavakoli H, Addolorato G, et al. Transcranial electrical and magnetic stimulation (tES and TMS) for addiction medicine: a consensus paper on the present state of the science and the road ahead. Neurosci Biobehav Rev. 2019;104:118-140.
4. Zangen A, Moshe H, Martinez D, et al. Repetitive transcranial magnetic stimulation for smoking cessation: a pivotal multicenter double-blind randomized controlled trial. World Psychiatry. 2021;20(3):397-404.
5. Koob GF, Volkow ND. Neurobiology of addiction: a neurocircuitry analysis. Lancet Psychiatry. 2016;3(8):760-773.
6. Bashir S, Yoo WK. Neuromodulation for addiction by transcranial direct current stimulation: opportunities and challenges. Ann Neurosci. 2016;23(4):241-245.
7. Thair H, Holloway AL, Newport R, Smith AD. Transcranial direct current stimulation (tDCS): a beginner’s guide for design and implementation. Front Neurosci. 2017;11:641.
8. Nitsche MA, Paulus W. Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J Physiol. 2000;527(Pt 3):633-639.
9. Bastani A, Jaberzadeh S. Differential modulation of corticospinal excitability by different current densities of anodal transcranial direct current stimulation. PLoS One. 2013;8(8):e72254.
10. Chen J, Qin J, He Q, Zou Z. A meta-analysis of transcranial direct current stimulation on substance and food craving: what effect do modulators have? Front Psychiatry. 2020;11:598.
11. Diana M, Raij T, Melis M, et al. Rehabilitating the addicted brain with transcranial magnetic stimulation. Nat Rev Neurosci. 2017;18(11):685-693.
12. Gorelick DA, Zangen A, George MS. Transcranial magnetic stimulation in the treatment of substance addiction. Ann N Y Acad Sci. 2014;1327(1):79-93.
13. Deng ZD, Lisanby SH, Peterchev AV. Electric field depth-focality tradeoff in transcranial magnetic stimulation: simulation comparison of 50 coil designs. Brain Stimul. 2013;6(1):1-13.
14. Levkovitz Y, Isserles M, Padberg F, et al. Efficacy and safety of deep transcranial magnetic stimulation for major depression: a prospective multicenter randomized controlled trial. World Psychiatry. 2015;14(1):64-73.
15. Goldstein RZ, Volkow ND. Dysfunction of the prefrontal cortex in addiction: neuroimaging findings and clinical implications. Nat Rev Neurosci. 2011;12(11):652-669.
16. Zilverstand A, Huang AS, Alia-Klein N, Goldstein RZ. Neuroimaging impaired response inhibition and salience attribution in human drug addiction: a systematic review. Neuron. 2018;98(5):886-903.
17. Jansen JM, Daams JG, Koeter MW, et al. Effects of non-invasive neurostimulation on craving: a meta-analysis. Neurosci Biobehav Rev. 2013;37(10 Pt 2):2472-2480.
18. Boggio PS, Sultani N, Fecteau S, et al. Prefrontal cortex modulation using transcranial DC stimulation reduces alcohol craving: a double-blind, sham-controlled study. Drug Alcohol Depend. 2008;92(1-3):55-60.
19. da Silva MC, Conti CL, Klauss J, et al. Behavioral effects of transcranial direct current stimulation (tDCS) induced dorsolateral prefrontal cortex plasticity in alcohol dependence. J Physiol Paris. 2013;107(6):493-502.
20. den Uyl TE, Gladwin TE, Wiers RW. Transcranial direct current stimulation, implicit alcohol associations and craving. Biol Psychol. 2015;105:37-42.
21. Wietschorke K, Lippold J, Jacob C, et al. Transcranial direct current stimulation of the prefrontal cortex reduces cue-reactivity in alcohol-dependent patients. J Neural Transm (Vienna). 2016;123(10):1173-1178.
22. Klauss J, Penido Pinheiro LC, Silva Merlo BL, et al. A randomized controlled trial of targeted prefrontal cortex modulation with tDCS in patients with alcohol dependence. Int J Neuropsychopharmacol. 2014;17(11):1793-1803.
23. Fregni F, Liguori P, Fecteau S, et al. Cortical stimulation of the prefrontal cortex with transcranial direct current stimulation reduces cue-provoked smoking craving: a randomized, sham-controlled study. J Clin Psychiatry. 2008;69(1):32-40.
24. Boggio PS, Liguori P, Sultani N, et al. Cumulative priming effects of cortical stimulation on smoking cue-induced craving. Neurosci Lett. 2009;463(1):82-86.
25. Fecteau S, Agosta S, Hone-Blanchet A, et al. Modulation of smoking and decision-making behaviors with transcranial direct current stimulation in tobacco smokers: a preliminary study. Drug Alcohol Depend. 2014;140:78-84.
26. Mondino M, Luck D, Grot S, et al. Effects of repeated transcranial direct current stimulation on smoking, craving and brain reactivity to smoking cues. Sci Rep. 2018;8(1):8724.
27. Perri RL, Perrotta D. Transcranial direct current stimulation of the prefrontal cortex reduces cigarette craving in not motivated to quit smokers: a randomized, sham-controlled study. Addict Behav. 2021;120:106956.
28. Batista EK, Klauss J, Fregni F, et al. A randomized placebo-controlled trial of targeted prefrontal cortex modulation with bilateral tDCS in patients with crack-cocaine dependence. Int J Neuropsychopharmacol. 2015;18(12):pyv066.
29. Coles AS, Kozak K, George TP. A review of brain stimulation methods to treat substance use disorders. Am J Addict. 2018;27(2):71-91.
30. Vanderhasselt MA, Allaert J, De Raedt R, et al. Bifrontal tDCS applied to the dorsolateral prefrontal cortex in heavy drinkers: influence on reward-triggered approach bias and alcohol consumption. Brain Cogn. 2020;138:105512.
31. Dinur-Klein L, Dannon P, Hadar A, et al. Smoking cessation induced by deep repetitive transcranial magnetic stimulation of the prefrontal and insular cortices: a prospective, randomized controlled trial. Biol Psychiatry. 2014;76(9):742-749.
32. Addolorato G, Antonelli M, Cocciolillo F, et al. Deep transcranial magnetic stimulation of the dorsolateral prefrontal cortex in alcohol use disorder patients: effects on dopamine transporter availability and alcohol intake. European Neuropsychopharmacol. 2017;27(5):450-461.
33. Terraneo A, Leggio L, Saladini M, et al. Transcranial magnetic stimulation of dorsolateral prefrontal cortex reduces cocaine use: a pilot study. European Neuropsychopharmaocl. 2015;26(1):37-44.
34. Liu X, Zhao X, Shen Y, et al. The effects of DLPFC targeted repetitive transcranial magnetic stimulation on craving in male methamphetamine patients. Clin Transl Med. 2020;10(2):e48.
35. Antonelli M, Fattore L, Sestito L, et al. Transcranial magnetic stimulation: a review about its efficacy in the treatment of alcohol, tobacco and cocaine addiction. Addict Behav. 2021;114:106760.
36. Song S, Zilverstand A, Gui W, et al. Effects of single-session versus multi-session non-invasive brain stimulation on craving and consumption in individuals with drug addiction, eating disorders or obesity: a meta-analysis. Brain Stimul. 2019;12(3):606-618.
37. Shen Y, Cao X, Tan T, et al. 10-Hz repetitive transcranial magnetic stimulation of the left dorsolateral prefrontal cortex reduces heroin cue craving in long-term addicts. Biol Psychiatry. 2016;80(3):e13-e14.
38. Jin L, Yuan M, Zhang W, et al. Repetitive transcranial magnetic stimulation modulates coupling among large-scale brain networks in heroin-dependent individuals: a randomized resting-state functional magnetic resonance imaging study. Addict Biol. 2022;27(2):e13121.
39. Liu X, Zhao X, Liu T, et al. The effects of repetitive transcranial magnetic stimulation on cue-induced craving in male patients with heroin use disorder. EBioMedicine. 2020;56:102809.
40. Pradhan B, Rossi G. Combining ketamine, brain stimulation (rTMS) and mindfulness therapy (TIMBER) for opioid addiction. Cureus. 2020;12(11):e11798.
41. Li X, Song GF, Yu JN, et al. Effectiveness and safety of repetitive transcranial magnetic stimulation for the treatment of morphine dependence: a retrospective study. Medicine (Baltimore). 2021;100(14):e25208.
42. Imperatore JP, McCalley DM, Borckardt JJ, et al. Non-invasive brain stimulation as a tool to decrease chronic pain in current opiate users: a parametric evaluation of two promising cortical targets. Drug Alcohol Depend. 2021;218:108409.
43. Sahlem GL, Caruso MA, Short EB, et al. A case series exploring the effect of twenty sessions of repetitive transcranial magnetic stimulation (rTMS) on cannabis use and craving. Brain Stimul. 2020;13(1):265-266.
44. Sahlem GL, Baker NL, George MS, et al. Repetitive transcranial magnetic stimulation (rTMS) administration to heavy cannabis users. Am J Drug Alcohol Abuse. 2018;44(1):47-55.
45. Prashad S, Dedrick ES, To WT, et al. Testing the role of the posterior cingulate cortex in processing salient stimuli in cannabis users: an rTMS study. Eur J Neurosci. 2019;50(3):2357-2369.
46. Kiluk BD, Fitzmaurice GM, Strain EC, Weiss RD. What defines a clinically meaningful outcome in the treatment of substance use disorders: reductions in direct consequences of drug use or improvement in overall functioning? Addiction. 2019;114(1):9-15.
47. Mahoney JJ 3rd, Hanlon CA, Marshalek PJ, et al. Transcranial magnetic stimulation, deep brain stimulation, and other forms of neuromodulation for substance use disorders: review of modalities and implications for treatment. J Neurol Sci. 2020;418:117149.
48. Rapinesi C, Curto M, Kotzalidis GD, et al. Antidepressant effectiveness of deep transcranial magnetic stimulation (dTMS) in patients with major depressive disorder (MDD) with or without alcohol use disorders (AUDs): a 6-month, open label, follow-up study. J Affect Disord. 2015;174:57-63.
49. Perrotta D, Perri RL. Mini-review: when neurostimulation joins cognitive-behavioral therapy. on the need of combining evidence-based treatments for addiction disorders. Neurosci Lett. 2022;777:136588.
50. Ma T, Sun Y, Ku Y. Effects of non-invasive brain stimulation on stimulant craving in users of cocaine, amphetamine, or methamphetamine: a systematic review and meta-analysis. Front Neurosci. 2019;13:1095.
51. Silvanto J, Pascual-Leone A. State-dependency of transcranial magnetic stimulation. Brain Topogr. 2008;21(1):1-10.
52. Silvanto J, Cattaneo Z, Battelli L, Pascual-Leone A. Baseline cortical excitability determines whether TMS disrupts or facilitates behavior. J Neurophysiol. 2008;99(5):2725-2730.
53. Krause B, Cohen Kadosh R. Not all brains are created equal: the relevance of individual differences in responsiveness to transcranial electrical stimulation. Front Syst Neurosci. 2014;8:25.
54. Chaieb L, Antal A, Paulus W. Gender-specific modulation of short-term neuroplasticity in the visual cortex induced by transcranial direct current stimulation. Vis Neurosci. 2008;25(1):77-81.
55. Rossi S, Hallett M, Rossini PM, Pascual-Leone A; Safety of TMS Consensus Group. Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Clin Neurophysiol. 2009;120(12):2008-2039.
56. Poreisz C, Boros K, Antal A, Paulus W. Safety aspects of transcranial direct current stimulation concerning healthy subjects and patients. Brain Res Bull. 2007;72(4-6):208-214.
57. Vigod S, Dennis CL, Daskalakis Z, et al. Transcranial direct current stimulation (tDCS) for treatment of major depression during pregnancy: study protocol for a pilot randomized controlled trial. Trials. 2014;15:366.
58. Brunoni AR, Amadera J, Berbel B, et al. A systematic review on reporting and assessment of adverse effects associated with transcranial direct current stimulation. Int J Neuropsychopharmacol. 2011;14(8):1133-1145.
59. Bari A, DeCisare J, Babayan D, et al. Neuromodulation for substance addiction in human subjects: a review. Neurosci Biobehav Rev. 2018;95:33-43.
60. Spagnolo PA, Goldman D. Neuromodulation interventions for addictive disorders: challenges, promise, and roadmap for future research. Brain. 2017;140(5):1183-1203.
61. Trojak B, Meille V, Achab S, et al. Transcranial magnetic stimulation combined with nicotine replacement therapy for smoking cessation: a randomized controlled trial. Brain Stimul. 2015;8(6):1168-1174.

Newsletter
Receive trusted psychiatric news, expert analysis, and clinical insights — subscribe today to support your practice and your patients.






