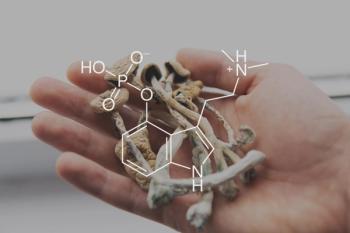
Positive Results From SKYLARK for Patients With Postpartum Depression
Zuranolone, an investigational oral drug being evaluated in women with postpartum depression, sees positive phase 3 study results.
Biogen and Sage Therapeutics announced positive results from the phase 3 SKYLARK study of
“Reducing suffering from
SKYLARK investigators evaluated 200 women who had a Hamilton Depression Rating Scale (HAMD-17) score of 26 or greater in a randomized, double-blind, placebo-controlled study evaluating the efficacy and safety of zuranolone 50 mg dosed once daily for 14 days compared to placebo. Participants were followed for up to 45 days. The study population included approximately 22% Black or African American women and 38% Hispanic or Latina women. Women treated with zuranolone 50 mg (n=98) demonstrated a statistically significant and clinically meaningful improvement in symptoms of depression at day 15—the primary endpoint.
“Postpartum depression is frequently under-recognized, and it can take time for women to be clinically diagnosed and treated,” said Katherine Dawson, MD, head of the Therapeutics Development Unit at Biogen. “Our hope is to be able to offer an innovative treatment option to potentially reduce the overwhelming impact
The study also met key secondary endpoints: Zuranolone 50 mg demonstrated statistically significant improvement in change from baseline in the Clinical Global Impression Severity (CGI-S) scale at day 15 as compared to placebo (zuranolone -2.2 vs. placebo -1.6, p= 0.0052). The CGI-S is a clinician-administered 7-point scale that rates the severity of an individual’s disease at the time of assessment.
“The positive outcomes of the SKYLARK Study are a critical step in our mission to help women suffering with postpartum depression find rapid relief from their depressive symptoms so they can get back to feeling like themselves,” said Barry Greene, chief executive officer at Sage. “Postpartum depression can be devastating for women and their families. If approved, zuranolone would be the first oral medication specifically indicated to treat PPD.”
Sage and Biogen have already begun a rolling submission for zuranolone in major depressive disorder (MDD) and aim to complete it in the second half of the year. An associated filing for zuranolone in PPD is planned for early next year.
Reference
1. Sage Therapeutics and Biogen announce that the phase 3 SKYLARK study of zuranolone in postpartum depression met its primary and all key secondary endpoints. Biogen. News release. June 1, 2022. Accessed June 2, 2022.