Publication
Article
Psychiatric Times
Medication-Assisted Psychotherapy: The Past and Future of Psychiatry
Author(s):
ART STOCK CREATIVE/AdobeStock

Psychiatric practice has increasingly relied on medication management rather than psychotherapy as a mainstay of treatment over the past few decades, despite the general recognition that pharmacology alone is insufficient for most patients. Combined pharmacological and psychosocial treatment, therefore, remains the optimal goal of psychiatry care. In addition, the growth of public interest in this area and the explosion of new research around psychedelics indicate an important place for combined treatments in the near future of clinical psychiatry. The questions for clinicians have become: How can we understand medication and psychotherapy and their interactive potential, and how can medication be understood and employed as a tool to enhance progress in the context of psychotherapy? To best answer these questions, we need a better understanding of the literature on combined medication-psychotherapy and medication-assisted psychotherapy.
Pharmacology and psychotherapy have been combined in recent literature in the use of fast-acting psychoactive compounds to augment an intensive session of psychotherapy. This strategy is known as medication-assisted psychotherapy (MAP), substance-assisted psychotherapy (SAPT), and psychedelic psychotherapy (PAP), depending on the medicine utilized and the context in which it is given. The literature lacks a clear consensus on the exact definition of these terms and what differentiates each term from the other.
For the sake of consistency, we will use MAP to describe any combination of medication and psychotherapy, including conventional psychotropics, such as selective serotonin reuptake inhibitors (SSRIs) when the goal of medication use is to facilitate and enhance the psychotherapy process, and we will use PAP to describe faster-acting mind-altering substances, ie, typical and atypical psychedelics (eg, 3,4-methylenedioxymethamphetamine [MDMA], ketamine, psilocybin, LSD, ibogaine), used during intensive psychotherapeutic sessions. Psychedelics are currently being investigated in clinical trials in conjunction with the delivery of a psychological intervention with the aim of treating specific psychiatric conditions.
Historical Background
Psychiatry has stood apart as a medical specialty because of the distinctive emphasis on psychotherapy as a treatment modality. In the middle decades of the 20th century, psychoanalysis was the dominant paradigm in American psychiatry and the main approach for the treatment of psychiatric disorders. The pharmacological revolution began in the 1950s and 1960s when pharmacological treatments for major psychotic, mood, and anxiety disorders were developed and popularized. In these early decades, however, pharmacology was commonly employed as an adjunct to facilitate psychotherapy, and this attitude is reflected in the pharmaceutical advertisements of the era.1 For example, an early ad for Elavil (amitriptyline) noted2:
The psychiatrist’s office still represents the setting in which the psychoanalytic process recognizes its fullest potential. Frequently, however, an antidepressant must be employed to foster a working therapeutic relationship. With effective symptomatic relief often provided by Elavil, depressed patients may be able to concentrate on underlying factors instead of somatic manifestations.
This attitude toward medication use in the context of psychotherapy began to change in the 1990s with the popularization of SSRIs, which, due to their improved tolerability, allowed primary care physicians to prescribe (typically without therapy). Simultaneously, the influence of managed care on US health care made integrated treatment less feasible. Since then, there has been a steady decline in the amount of psychotherapy offered by psychiatrists. A 2021 study found that between 1996 and 2016, the percentage of psychiatric visits involving psychotherapy had decreased by half, dropping to 21.6% of patient visits in 2016.3 This number is likely to have dropped even further since then.
Interestingly, psychiatric training has protected its insistence upon psychotherapy as a competency for resident trainees. Many psychiatrists still identify as psychotherapists, and pride themselves on their mastery of both psychological and pharmacological treatment approaches. Psychiatric clinicians have a uniquely comprehensive capacity in the treatment of psychiatric patients, as they are the only group of mental health care professionals authorized to provide integrative pharmacotherapy and psychotherapy.
As readers know, a fully integrated approach, in which a single clinician offers both pharmacotherapy and psychotherapy, has several advantages, including enhanced continuity of care, improved communication, coordination between treatment modalities, and increased convenience for patients. This integrated approach also allows for a comprehensive understanding of the patient’s condition and the ability to tailor treatment to individual needs.
However, there are also potential disadvantages to this approach, which have likely contributed to its decline. Clinicians may face challenges in maintaining proficiency in both pharmacotherapy and psychotherapy due to the extensive training required for each. Additionally, time constraints may limit the depth and quality of each treatment modality, and financial pressures may incentivize psychiatrists to focus more of their time on medication management alone.
Historical Insights
The use of psychoactive compounds during psychotherapy has an extensive historical basis in psychiatry going at least as far back as the 1930s when William J. Bleckwenn, MD, started conducting experiments with subanesthetic doses of amylobarbitone in combination with analytic techniques.4 Bleckwenn demonstrated the potential of drug-assisted interviews in psychiatry and laid the foundation for further advancements in the field of barbiturate-assisted interviews and their application in psychiatric treatment. During World War II, the use of barbiturate-assisted interviews experienced a significant surge. The technique was particularly employed to facilitate abreaction, the release of previously repressed emotions, especially in the treatment of posttraumatic stress disorders (PTSDs). The enthusiasm for barbiturate-assisted interviews continued in civilian practice after the war, via various techniques including prolonged narcosis and narco-hypnoanalysis. However, by the early 1960s, the technique began to decline, mostly due to concerns about barbiturate dependence, the introduction of new psychiatric medications such as chlorpromazine and tricyclic antidepressants, and the popularity of hallucinogenic drugs (eg, LSD).4
Psychodynamic Psychopharmacology
Psychodynamic psychopharmacology is an approach that seeks to integrate the principles of psychodynamic theory with pharmacology. This area of research has emerged as an important approach to mental health treatment and has played an important role in the development of combined pharmacological and psychotherapeutic interventions. Psychodynamic psychopharmacology recognizes the role of meaning, psychosocial factors, and interpersonal dynamics in treatment-refractory mental illnesses. It highlights that psychosocial factors and the patient-doctor relationship are often overlooked in psychiatric treatment, leading to treatment resistance and limited outcomes. This is achieved by recognizing the importance of the integration of biological psychiatry and psychodynamic insights (Table 1). By incorporating psychodynamic principles into psychopharmacological practice, psychiatrists can better understand and address the complex psychological factors that influence treatment outcomes. This approach enhances the integration of meaning and biology, leading to more effective and patient-centered care for individuals with treatment-resistant mental illnesses.5,6
Table 1. The 6 Primary Principles of Psychodynamic Psychopharmacology

Combined Treatment
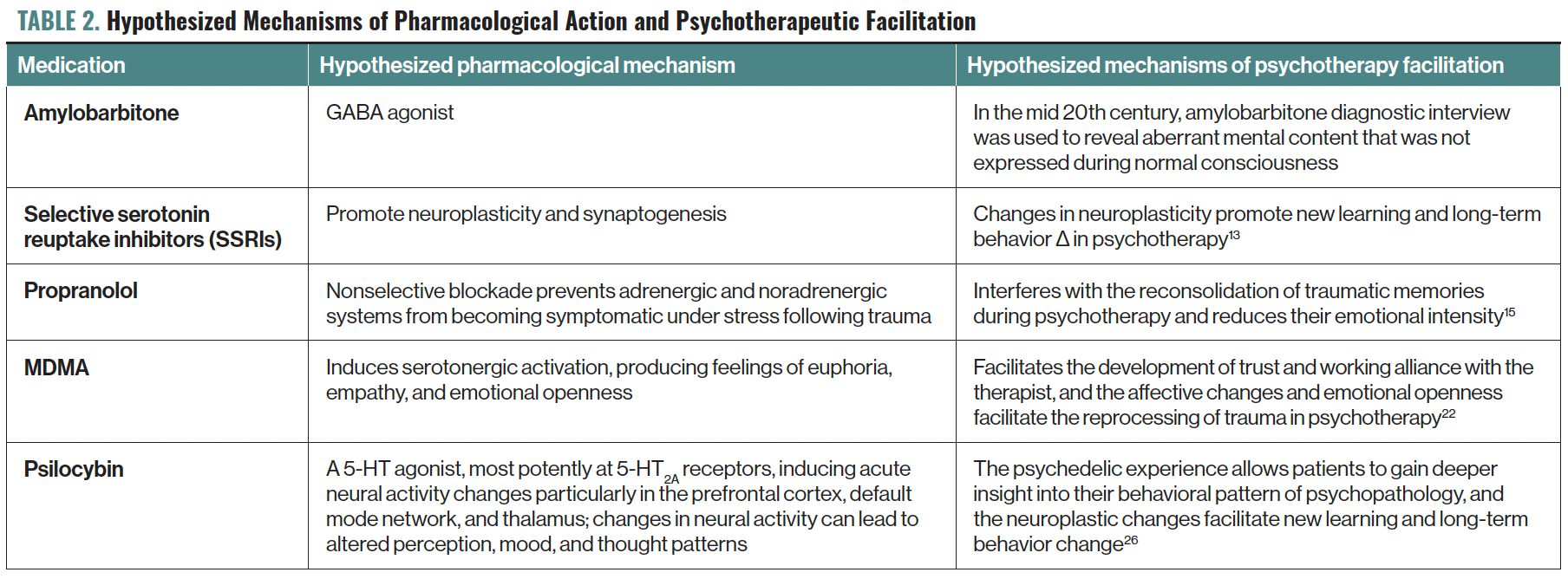
Comparative efficacy trials over the past few decades have provided evidence that a combination of psychotherapy with psychopharmacology together are more efficacious than either alone for a variety of diagnoses,7-10 with psychotherapy and medication differing in treatment efficacy for various target symptoms, having different rates of action, or enhancing duration of effective treatment.11,12 Moreover, because contemporary hypotheses around the mechanism of action of antidepressants center on neuroplasticity,13 this again suggests a vital role for psychosocial treatments to provide optimal learning environment for, to use a common metaphor, “rewiring of the brain” (Table 2). Thus, the plausibility of synergistic independent complementary effects of combined medication and psychotherapy is supported by clinical and neuroscientific literature.
Table 2. Hypothesized Mechanisms of Pharmacological Action and Psychotherapeutic Facilitation
Propranolol-Assisted Psychotherapy
The use of propranolol in conjunction with psychotherapy for PTSD began gaining attention after research in the 1980s postulated that epinephrine and norepinephrine play a role in formation of traumatic memories in PTSD.14
A β-blocker, propranolol acts as a competitive antagonist to inhibit catecholamines, so it was hypothesized that it might have therapeutic efficacy for the prevention and treatment of PTSD. Research investigating the use of propranolol has primarily focused on 2 main indications:
1. Use after the immediate experience of traumatic events to prevent consolidation of traumatic memories.
2. Given in combination with trauma therapy to reactivate traumatic memories. Propranolol was theorized to interfere with the reconsolidation process and weaken the emotional intensity associated with traumatic memories.15
Clinical studies have been conducted to investigate the efficacy of propranolol-assisted psychotherapy for PTSD. These studies involved administering propranolol prior to or after psychotherapeutic sessions, such as exposure therapy or cognitive-behavioral therapy. One of the earliest studies investigated the effects of propranolol in reducing the intensity of traumatic memories during therapy sessions for Vietnam combat veterans with chronic PTSD. The researchers found propranolol administration prior to therapy sessions led to a significant reduction in subjective distress associated with traumatic memories.16 Although a 2018 systematic review did not show any significant evidence that propranolol was effective for traumatic memory disruption in patients with PTSD, likely due to small sample sizes and heterogeneity of dosages as limitations,17 another study showed that the combination of propranolol and reconsolidation blockade (RRB) therapy led to a significant reduction in PTSD symptoms compared with placebo plus RRB therapy, indicating a potential synergistic effect of the combined treatment approach.18
MDMA
Recently MDMA has been investigated as a treatment for PTSD in the context of psychotherapy, with the US Food and Drug Administration granting breakthrough therapy designation for MDMA-assisted therapy for PTSD in 2017. The first phase 3 findings were published in 2021 with promising results.19 In the study, participants with severe PTSD were randomly assigned to receive either manualized therapy with MDMA (n=46) or with placebo (n = 44) at 1 of 15 study sites across the United States, Canada, and Israel. Participants underwent three 8-hour experimental sessions spaced 4 weeks apart with a single divided dose of 80 to 180 mg MDMA or placebo, in addition to 3 preparatory and 9 integrative therapy sessions.
In the first experimental session, participants received an initial dose of 80 mg followed by a supplemental half-dose of 40 mg, 1.5 to 2.5 hours later. In the second and third experimental sessions, participants received an initial dose of 120 mg followed by a supplemental half-dose of 60 mg. Each experimental session was followed by three 90-minute integrative sessions spaced 1 week apart to allow participants to incorporate their experiences. At the primary end point at 18 weeks, 67% of participants in the MDMA group no longer met diagnostic criteria for PTSD, compared with 32% of participants in the placebo group.19 Positive results from a second phase 3 trial have also been reported.20
MDMA is thought to work by reducing the emotional intensity associated with traumatic memories by reconsolidating traumatic memories during psychotherapy in a way that they are stored with less excitatory activation.21 MDMA has also been noted to increase feelings of trust, safety, and empathy, possibly mediated via release of oxytocin and serotonin, which can improve the therapeutic alliance between the therapist and individual.22,23
Psilocybin
Although psilocybin has been studied in conjunction with psychotherapy for several psychiatric diagnoses including nicotine dependence, alcohol dependence, obsessive-compulsive disorder, cluster headache, cancer-related existential distress, and anxiety disorders, much of the research has focused on the treatment of patients with a diagnosis of treatment resistant depression.23
Several phase 2 trials have shown the potential efficacy of psilocybin-assisted psychotherapy for the treatment of patients with treatment resistant depression and, in 2018, the FDA granted breakthrough therapy designation for psilocybin-assisted therapy for treatment-resistant depression. One of the phase 2 studies involved 24 patients with major depressive disorder who received either a single dose of psilocybin or a placebo in a therapeutic setting and were followed for 4 weeks after treatment.24 The results showed that psilocybin-assisted therapy led to significant reductions in depression symptoms as measured by the Hamilton Depression Rating Scale (HAM-D). At 1 week after treatment, the mean HAM-D score was significantly lower in the psilocybin group compared with the placebo group (8.0 vs 14.4). At 4 weeks post treatment, the psilocybin group still showed a significant reduction in mean HAM-D scores compared with baseline (10.9 vs 22.8), whereas the placebo group did not (19.3 vs 21.7). A prospective 12-month follow-up found that psilocybin-assisted therapy resulted in significant improvements in depressive symptoms at 1 week, 4 weeks, and 12 months after treatment, with the mean HAM-D scores decreasing from 22.8 at baseline to 8.0 at 1 week, 8.5 at 4 weeks, and 9.8 at 12 months after treatment.25
Additional Considerations
As opposed to medication management outside therapeutic context, there is an added importance on the type of experience during the PAP sessions. Specifically, mystical experiences, meaningful insights, and belief changes have frequently been cited as predictive of positive outcomes of psychedelic trials because they enable individuals to transcend their usual patterns of thinking, feeling, and acting.26,27 These experiences can increase insightfulness, introspection, divergent thinking, and mindfulness-related capacities; reduce avoidance; and induce emotional breakthrough experiences that can help resolve existential distress.27
In PAP, set and setting is thought to have a large impact on the outcomes of the therapies. Set refers to the individual’s mind-set, or the internal attitudes, expectations, and psychological state that they bring into the therapeutic experience. Setting, on the other hand, refers to the physical, social, and cultural environment where the therapy takes place. It covers the comfort and safety of the physical surroundings, the presence and demeanor of the therapist(s), and the broader societal and cultural attitudes toward psychedelics and mental health. A supportive, calm, and comfortable setting is essential to facilitate a positive therapeutic experience.
Neurophysiological and Psychological Views
The integrated use of medications to facilitate psychological recovery breaks down the dichotomy between the neurophysiological and psychological views. Mental processes, after all, are the product of physical brain activities. Experiences and environments can produce long-standing neurophysiological changes, recognized by phenomena such as neuroplasticity and epigenetics.28 Psychiatric medications instigate a cascade of neurotransmission involving key neurochemicals such as serotonin, dopamine, norepinephrine, glutamate, and oxytocin. These neurochemical alterations function as potent facilitators of neuroplasticity, serve to attenuate amygdala reactivity during emotional processing, and instigate reductions in the activity of the default mode network. The ensuing psychological alterations are multifaceted, encompassing an attenuation of fear responses and feelings of shame, augmented emotional empathy, enhanced processing of traumatic memories, and a marked increase in openness and interpersonal trust.29 Neurophysiological activity underlies psychological experiences, whereas psychological factors influence neurophysiological functioning. Recognizing their interdependence is crucial for a comprehensive understanding of mental health and a convergence toward a common understanding.
Concluding Thoughts
A substantial body of literature supports the efficacy of combining medications, illustrated in this narrative review by conventional medications such as SSRIs and propranolol as well as psychoactive compounds such as MDMA and psilocybin, with psychotherapeutic interventions for treatment of many psychiatric conditions. MAP recovers and reintroduces an emphasis on using the experiential aspect of psychotropics to enhance progress in psychotherapy. Many methodological challenges remain regarding the clinical study of psychedelics (see van Elk and Fried30) but available results are promising.
Research evidence continues to indicate the additional benefits of the synergistic use of medication with psychotherapy. As opposed to the currently dominant model of medication as the primary treatment modality, many of these approaches emphasize the importance of the experience itself and point to the ways in which medications alter the neurobiology to facilitate neuroplasticity, learning, and behavioral modifications.
It is the responsibility of all psychiatric clinicians to familiarize themselves with integrative treatment for the benefit of their patients. Our expertise in both the pharmacological and psychological aspects of patient care will be key to providing the best care possible.
Dr Malidelis is PGY4 at Case Western Reserve University, University Hospitals Cleveland Medical Center, Adult Psychiatry Residency Program, Cleveland, Ohio. Dr Hunt is assistant program director, Case Western Reserve University, University Hospitals Cleveland Medical Center, Adult Psychiatry Residency Program. Dr Aftab is clinical assistant professor, Department of Psychiatry, Case Western Reserve University School of Medicine. He also is a member of the Psychiatric Times Editorial Board. All authors report no conflicts of interest and have no disclosures to report.
References
1. Neill JR. A social history of psychotropic drug advertisements. Soc Sci Med. 1989;28(4):333-338.
2. Elavil (amitriptyline). Accessed December 6, 2023. https://slatestarcodex.com/blog_images/psychad16.png
3. Tadmon D, Olfson M. Trends in outpatient psychotherapy provision by U.S. psychiatrists: 1996–2016. Am J Psychiatry. 2022;179(2):110-121.
4. Patrick M, Howells R. Barbiturate-assisted interviews in modern clinical practice. Psychol Med. 1990;20(4):763-765.
5. Mintz D, Belnap B. A view from Riggs: treatment resistance and patient authority—III. What is psychodynamic psychopharmacology? An approach to pharmacologic treatment resistance. J Am Acad Psychoanal Dyn Psychiatry. 2006;34(4):581-601.
6. Mintz D. Psychodynamic Psychopharmacology: Caring for the Treatment-Resistant Patient. American Psychiatric Association Publishing; 2022.
7. Keller MB, McCullough JP, Klein DN, et al. A comparison of nefazodone, the cognitive behavioral-analysis system of psychotherapy, and their combination for the treatment of chronic depression. N Engl J Med. 2000;342(20):1462-1470.
8. Dixon LB, Dickerson F, Bellack AS, et al. The 2009 schizophrenia PORT psychosocial treatment recommendations and summary statements. Schizophr Bull. 2010;36(1):48-70.
9. Mavissakalian M. Combined behavioral therapy and pharmacotherapy of agoraphobia. J Psychiatr Res. 1993;27(suppl 1):179-191.
10. Cuijpers P, Noma H, Karyotaki E, et al. A network meta-analysis of the effects of psychotherapies, pharmacotherapies and their combination in the treatment of adult depression. World Psychiatry. 2020;19(1):92-107.
11. Boschloo L, Bekhuis E, Weitz ES, et al. The symptom specific efficacy of antidepressant medication vs. cognitive behavioral therapy in the treatment of depression: results from an individual patient data meta-analysis. World Psychiatry. 2019;18(2):183-191.
12. Boschloo L, Hieronymus F, Cuijpers P, et al. Clinical response to SSRIs relative to cognitive behavioral therapy in depression: a symptom specific approach. World Psychiatry. 2022;21(1):152-153.
13. Casarotto PC, Girych M, Fred SM, et al. Antidepressant drugs act by directly binding to TRKB neurotrophin receptors. Cell. 2021;184(5):1299-1313.e19.
14. Pitman RK. Post-traumatic stress disorder, hormones, and memory. Biol Psychiatry. 1989;26(3):221–223.
15. Young C, Butcher R. Propranolol for Post-Traumatic Stress Disorder: A Review of Clinical Effectiveness. Canadian Agency for Drugs and Technologies in Health; March 18, 2020. Accessed December 6, 2023. https://www.ncbi.nlm.nih.gov/books/NBK562942/
16. Pitman RK, Sanders KM, Zusman RM, et al. Pilot study of secondary prevention of posttraumatic stress disorder with propranolol. Biol Psychiatry. 2002;51(2):189-192.
17. Raut SB, Canales JJ, Ravindran M, et al. Effects of propranolol on the modification of trauma memory reconsolidation in PTSD patients: a systematic review and meta-analysis. J Psychiatr Res. 2022;150:246-256.
18. Brunet A, Saumier D, Liu A, et al. Reduction of PTSD symptoms with pre-reactivation propranolol therapy: a randomized controlled trial. Am J Psychiatry. 2018;175(5):427-433.
19. Mitchell JM, Bogenschutz M, Lilienstein A, et al. MDMA-assisted therapy for severe PTSD: a randomized, double-blind, placebo-controlled phase 3 study. Nat Med. 2021;27(6):1025-1033.
20. Mitchell JM, Ot’alora G M, van der Kolk B, et al. MDMA-assisted therapy for moderate to severe PTSD: a randomized, placebo-controlled phase 3 trial. Nat Med. 2023;29(10):2473-2480.
21. Jerome L, Feduccia AA, Wang JB, et al. Long-term follow-up outcomes of MDMA-assisted psychotherapy for treatment of PTSD: a longitudinal pooled analysis of six phase 2 trials. Psychopharmacology (Berl). 2020;237(8):2485–2497.
22. Singleton SP, Wang JB, Mithoefer M, et al. Altered brain activity and functional connectivity after MDMA-assisted therapy for post-traumatic stress disorder. Front Psychiatry. 2023;13:947622.
23. Hodge AT, Sukpraprut-Braaten S, Narlesky M, Strayhan RC. The use of psilocybin in the treatment of psychiatric disorders with attention to relative safety profile: a systematic review. J Psychoactive Drugs. 2023;55(1):40-50.
24. Davis AK, Barrett FS, May DG, et al. Effects of psilocybin-assisted therapy on major depressive disorder: a randomized clinical trial. JAMA Psychiatry. 2021;78(5):481-489.
25. Gukasyan N, Davis AK, Barrett FS, et al. Efficacy and safety of psilocybin-assisted treatment for major depressive disorder: prospective 12-month follow-up. J Psychopharmacol. 2022;36(2):151-158.
26. Yaden DB, Griffiths RR. The subjective effects of psychedelics are necessary for their enduring therapeutic effects. ACS Pharmacol Transl Sci. 2020;4(2):568-572.
27. Krediet E, Bostoen T, Breeksema J, et al. Reviewing the potential of psychedelics for the treatment of PTSD. Int J Neuropsychopharmacol. 2020;23(6):385-400.
28. Gräff J, Mansuy IM. Epigenetic codes in cognition and behaviour. Behav Brain Res. 2008;192(1):70-87.
29. Thal SB, Bright SJ, Sharbanee JM, et al. Current perspective on the therapeutic preset for substance-assisted psychotherapy. Front Psychol. 2021;12:617224.
30. van Elk M, Fried EI. History repeating: guidelines to address common problems in psychedelic science. Ther Adv Psychopharmacol. 2023;13:20451253231198466.

Newsletter
Receive trusted psychiatric news, expert analysis, and clinical insights — subscribe today to support your practice and your patients.